Lipoprotein (a) Assay
_KIT.jpg)
|
| Product Features
Diazyme's Lipoprotein (a) is a dual vial, liquid stable reagent system which provides reliable results. The assay method requires only 6 μL of serum sample and is completed in less than 10 minutes. Diazyme's Lipoprotein (a) assay has excellent correlation with existing commercial products with a correlation coefficient of 0.9868, slope of 0.9828, and y intercept of 1.0033. The linear range extends up to 100 mg/dL and has Within-Run and Between-Run precision CV values ≤ 3.7%. |
||
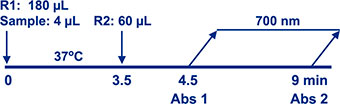 Assay Principle Assay Principle
The Diazyme Lipoprotein (a) Assay is based on a latex enhanced immunoturbidimetric assay. Lp(a) in the sample binds to specific anti- Lipoprotein (a) antibody, which is coated on latex particles, and causes agglutination. The degree of the turbidity caused by agglutination can be measured optically and is proportional to the amount of Lipoprotein (a) in the sample. |
||
| Intended use
The Diazyme Lipoprotein (a) Assay is intended as a latex particle enhanced immunoturbidimetric assay for the in vitro quantitative determination of Lp(a) concentration in human serum or plasma on Clinical Chemistry Systems. The measurement of Lipoprotein (a) is useful in evaluating lipid metabolism disorders and assessing atherosclerotic cardiovascular diseases in specific populations, when used in conjunction with clinical evaluation. For in vitro diagnostic use only. |
||
| Ordering Information | ||
| Description | Catalog No. | Packaging |
| Lipoprotein (a) Assay | DZ131B-KY1 | R1: 1 x 60 mL R2: 1 x 20 mL |
| Lipoprotein (a) Calibrator Set | DZ131B-CAL | Cal: 5 x 1 mL |
| Lipoprotein (a) Control Set | DZ131B-CON | Con: 2 x 1 mL |
| Regulatory Status
|
||



