Lithium Assay

 |
| Product Features
Diazyme's Lithium Assay provides a convenient, stable, non-caustic low cost alternative to expensive ISE and highly caustic alkaline Lithium methods. Diazyme's Lithium Assay demonstrates an excellent correlation with ISE methods and offers excellent precision CV% of <5% and a measuring range from 0.19 mmol/L – 3.0 mmol/L. The assay is not interfered by the following substances at indicated concentrations: Na+ 200 mM, NH4+ 0.5 mM, Ca2+ 4.0 mM, Mg2+ 2.0 mM, ascorbic acid 5.0 mM, 0.25 mM Zn2+, 0.25 mM Fe3+, 0.25 mM Cu2+, 10 mM K+, Triglycerides 1000 mg/dL, conjugated bilirubin 20 mg/dL and unconjugated bilirubin 45 mg/ dL, Ascorbic Acid 5 mM and hemoglobin 500 mg/dL. |
|||
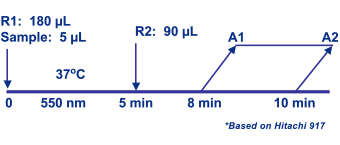 Assay Principle Assay Principle
Lithium is determined spectrophotometrically through a kinetic coupled enzyme assay system involving Diazyme's proprietary phosphatase1 whose activity is sensitive to lithium (IC50=0.1mM). Through enzymatic coupling, the phosphatase substrate is converted to hypoxanthine by a series of enzymatic reactions to generate uric acid and hydrogen peroxide (H2O2). H2O2 generated reacts with N-Ethyl-N-(2-hydroxy-3-sulfopropyl)-3-methylaniline (EHSPT) and 4-aminoantipyrine (4-AA) in the presence of peroxidase (POD) to form a quinone dye which has maximal absorbance at 556 nm. The rate of the quinine dye formation is inversely proportional to the concentration of lithium in serum samples. |
|||
| Intended use
Diazyme Lithium Enzymatic Assay kit is for quantitative in vitro determination of lithium in human serum. Measurements of Lithium are carried out essentially to ensure that proper drug dosage is administered in the treatment of patient suffering from bipolar disorder and to avoid toxicity. |
|||
| Product | Catalog Number | Format | Method |
| Kit | DZ116B | R1/R2 (Dual Vial Liquid Stable) | Dual Vial Liquid Stable, Enzymatic |
| Calibrator | DZ116B-Cal | Cal: 3 Level | |
| Control | DZ116B-Con | Con: 2 Level | |
| Regulatory Status
|
|||



